Maintaining recreational pond health requires an understanding of various water quality parameters and how they interact with the living and non-living components of a pond. Properly managing water quality can mean the difference between a severe summer algal bloom that depletes the pond of oxygen, or a productive summer of fishing with friends and family. In addition, water quality can play a role in effective aquatic herbicide application, fertilization, and more. A proactive approach to pond management is one that will yield the best results.
Measuring Water Quality
Analyzing water quality can be carried out through various means. There are several aquaculture test kits that can be purchased online for a reasonable price and some pond management companies may be able to provide this service. Additionally, the Agriculture Service Lab through Clemson Regulatory Services has several options to help analyze and interpret water quality in recreational ponds. The Irrigation Water analysis can identify levels of common components of water, such as magnesium, calcium, pH, heavy metals, and much more. Even better, Clemson has published an easy to use document, Interpreting Irrigation Water Quality Reports, to help identify areas of the pond that require adjustments. Whether landowners are monitoring their own pond, consulting with a pond management company, or using Clemson Extension resources, here are a few important elements to keep in mind.
Dissolved Oxygen

Figure 1: A dragonfly nymph has crawled onto the underside of a mushroom and the adult has recently emerged. The adult pauses to dry its wings before the nuptial flight.
Karen Jackson, ©2020, Clemson University
One of the most important water quality paraments for any water body is dissolved oxygen. Dissolved oxygen is essential to support a healthy fish community and is needed by benthic macroinvertebrates that serve as food for fish. Benthic macroinvertebrates are organisms that do not have a backbone but are able to be seen without the use of a microscope. One example often found in ponds is a dragonfly nymph, which begins its life in the water prior to emerging as an adult (figure 1). Oxygen is introduced into the system via photosynthesis from algae and phytoplankton, mixing from wind and natural diffusion from the atmosphere. Aeration devices such as bottom diffusers can also help to disperse oxygen throughout the system, while also preventing pond turnovers. Many pond owners consider the use of fountains for oxygen dispersion, but for deeper ponds, surface fountains may only serve as an aesthetic component of the pond and are unable to oxygenate the full water column. Measured in parts per million (ppm) or milligrams per liter (mg/L), dissolved oxygen levels in a pond below 3 mg/L will stress aquatic species and make them more susceptible to disease. Dissolved oxygen concentrations vary throughout a 24-hour period due to photosynthesizing plants and algae. Whereas photosynthesis will increase concentrations during the day, plant respiration at night will lower dissolved oxygen (figure 2). This is important to keep in mind if developing a sampling regime for a pond. Temperature can also affect dissolved oxygen, as cooler water is able to hold onto oxygen molecules better than warmer water (figure 3).

Figure 2: Dissolved oxygen changes fluctuate throughout a 24-hour period, with the highest concentrations during the afternoon and lowest at night.
Karen Jackson, ©2020, Clemson University
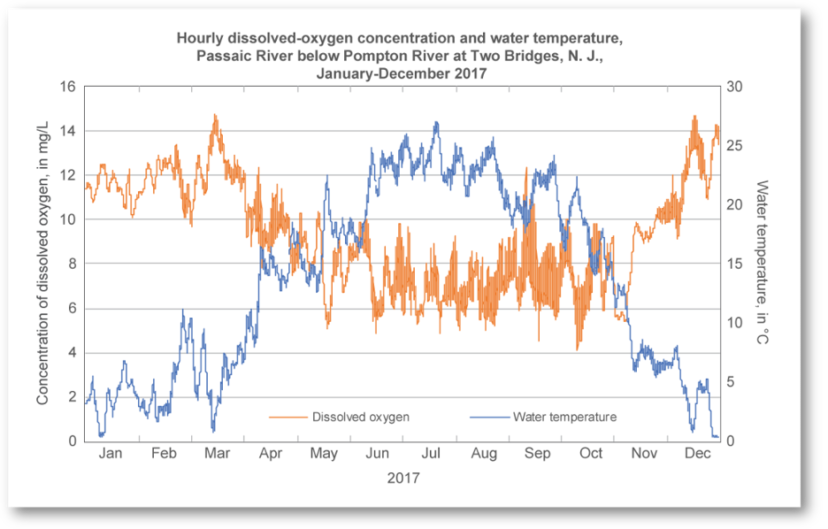
Figure 3: Dissolved oxygen concentration (mg/L) are inversely proportionate to water temperature.
Photo credit: USGS
Nutrients are important in pond ecosystems to support a healthy fish and aquatic organism population (figure 4). However, an excessive amount of nutrients in a pond, also known as eutrophication, can result in large algal blooms and is one of the most likely causes of low dissolved oxygen. Phosphorus and nitrogen are found in stormwater runoff and can enter the pond easily, especially where there is no existing shoreline buffer, see HGIC 1855, Shorescaping Freshwater Shorelines. Sources of nutrients in runoff include pet and livestock waste, excess fertilizers, and lawn debris. Excess nitrogen and phosphorus cause an increase in algal growth, which can result in a loss of dissolved oxygen in two different ways. If algae proliferate to a point at which visibility is reduced to less than 12 inches, a dramatic reduction in oxygen will occur. If the algae then die and begin to decompose, bacteria will respire and reduce the amount of dissolved oxygen in the pond. The South Carolina Department of Health and Environmental Control has published Regulation 61-68: Water Classifications and Standards to manage and protect waters of the state. This document provides guidance on nutrient values in waterbodies in different geographic locations within the state.
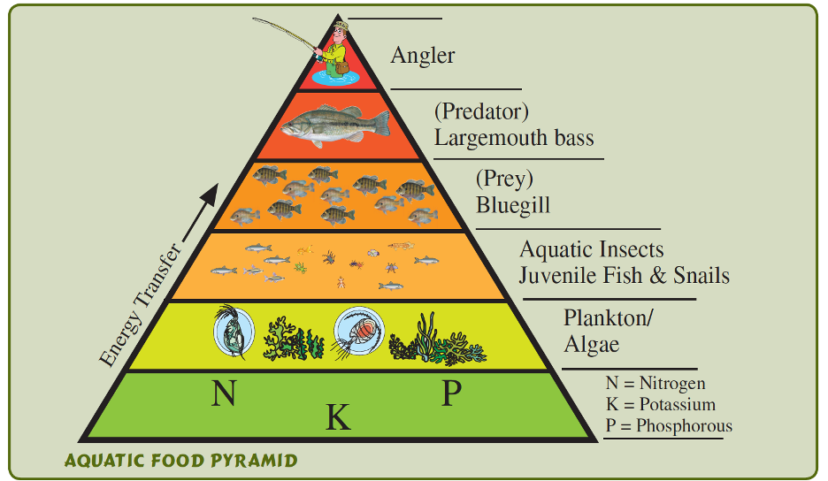
Figure 4: Nutrients are essential to support a healthy community of aquatic organisms.
Photo credit: SC Department of Natural Resources.

Figure 5: A secchi disk is an alternating black and white object that is lowered into the water column to estimate turbidity and clarity, and to determine if fertilization is needed.
Karen Jackson, ©2020, Clemson University
In contrast, pond owners may initiate a fertilization program specifically to support fish production. If this is the case, an inorganic fertilizer is applied in March or April until a secchi disk is not visible within the first 18 inches under the water (figure 5).
Prior to fertilizing ponds, ownersmust consider any downstream impacts from an increase in nutrients. Nutrient-rich water leaving the pond can have adverse impacts on receiving waterways. For more on this, visit HGIC 1710 Fertilizing Recreational Fish Pond. The Southern Regional Aquaculture Center also has guidelines for pond owners in their publication No. 0469, Fertilizing Fish Ponds. It is important to note that stormwater ponds should never be fertilized, as it will increase the likelihood of an algal bloom. These ponds are designed to capture and treat stormwater runoff, which is already assumed to be nutrient-rich.
Water Clarity
Water clarity can also be a contributing factor to poor pond water quality and can be influenced by increases in sediment, total suspended solids, or total dissolved solids. Each of these can adversely affect water quality and aquatic organisms. Sediment, for instance, can become abrasive to the gills of aquatic organisms, limit sunlight for plant growth, and provide a pathway for other pollutants to enter the pond. Pollutants such as herbicide, bacteria, and fertilizer can adhere to sediment particles as they enter the way. Sediment often enters the pond by way of erosion. Erosion can be prevented by establishing a native buffer around the perimeter of the pond. This will stabilize the shoreline and capture any sediment found in stormwater runoff. Additionally, stabilize bare areas in the upland and consider the use of stormwater management practices such as rain gardens, bioswales, and rainwater harvesting to help manage runoff entering the pond.
Total suspended solids, or TSS, is the measure of all the solid, undissolved material in the water. This material comes from erosion, runoff, suspended sediments from the stream bottom, and algal blooms. High TSS can increase the temperature by attracting and absorbing sunlight, while also disrupting photosynthesis. This can then lead to lowered dissolved oxygen levels. Similar to sediment, suspended solids can become abrasive to fish and benthic macroinvertebrate gills, hindering respiration.
Turbidity is an estimate of the total suspended solids in water based on the amount of light scattered by particles. While this is not a direct measure of TSS in water, it can give a pond owner an estimate of water clarity. An easy way for a pond owner to estimate turbidity is by using a secchi disk. A secchi disk is a black and white disk that is lowered into the water until it is no longer visible. Ideally, the disk will be visible within the first 18 inches. More information on this can be found in HGIC 1710 Fertilizing Recreational Fish Ponds. Gypsum (CaSO4) can be added to turbid ponds and is widely available at farm supply stores. Gypsum works by attracting clay particles together, which then eventually settle to the bottom of the pond.
pH
A pond’s pH is the concentration of hydrogen ions in the water, or how acidic or basic the water is. The scale ranges from 0 (acidic) to 14 (basic), with 7 representing neutral. The optimum range for pond water for fish health is 6.5-9.0, though they can survive in a range of 4.5 to 9. Vertebrates, including fish, have an average blood pH of 7.4 and are greatly affected when the pH of water is vastly different. Also, when the pH changes rapidly, fish can become stressed. The alkalinity of the pond can help prevent these unexpected swings in pH by serving as a buffer. Similar to dissolved oxygen, pH is affected by changes in photosynthesis and respiration throughout the day. For instance, nighttime respiration increases carbon dioxide in the water, which produces carbonic acid and lowers the pH, making the water slightly more acidic. pH is most accurately measured in the field, with the use of test strips or probe, as it is only accurate for 15 minutes after sample collection.
Alkalinity
The alkalinity of a pond is its ability to buffer against fluctuations in pH and neutralize the acid. When testing for alkalinity, measure bases like carbonates and bicarbonates. This is expressed in parts per million (ppm) of calcium carbonate, or CaCo3. Ideally, the alkalinity is near 100 ppm, but 50 to 200 ppm are acceptable ranges. A reading of 20 ppm or less may indicate a need to add agricultural limestone, especially if the plan is to fertilize the pond. An alkalinity of less than 20 ppm will cause the fertilizer to settle out in the pond bottom, rendering it useless to the water column. A Special Irrigation Test through the Clemson University Agricultural Service Laboratory can help to determine this. Total alkalinity is calculated by converting the results for HCO3 on this irrigation water analysis.
_____ (HCO3) X 61 = _____ Total Alkalinity (mg/L)
Hardness
Often confused with alkalinity, hardness is the measurement of divalent ions, specifically salts with two positive charges, mainly magnesium (Mg2+) and calcium (Ca2+). Whether the pond has hard or soft water is determined by the source of water and surrounding geology. For example, ponds in limestone, which is a calcium carbonate-rich rock, will have harder water. Similar to alkalinity, hardness levels that are too high or too low can cause herbicide treatments to be ineffective. Magnesium and calcium are essential elements for fish growth, such as scale and bone formation and molting in crustaceans. Salts in the body also help with osmoregulation that supports nerve and muscle function. Similar to alkalinity, hardness should be no less than 20 mg/L, but ideally, 50 to 200 mg/L and can be adjusted with agricultural limestone. The Special Irrigation Test through Clemson will report values for calcium and magnesium, and the follow conversion will identify total hardness:
(_____ Ca X 2.497) + (_____ Mg X 4.118) = ____________ Total Hardness (mg/L)
pH, alkalinity, and hardness all can play a role in herbicide or algaecide application. Pond owners will need to test all three components in their pond prior to any application. For instance, when the alkalinity is too low, it can cause the copper to become toxic to fish instead of an efficient algal treatment. Conversely, if the alkalinity is too high, the copper may be ineffective. Herbicide and algae labels will give additional information that should be followed precisely.
Temperature
As mentioned above, the temperature can most strongly influence dissolved oxygen, with colder water holding more oxygen relative to warmer water. The most dramatic effect of this is when spring and summer roll around and cause pond stratification in water deeper than six to eight feet. Water at the bottom of the pond, or hypolimnion, is colder and denser, with lower dissolved oxygen concentrations. Water on the surface, or epilimnion, is the warmest and least dense, with higher amounts of dissolved oxygen. The hypolimnion and epilimnion are separated by the thermocline, a marked gradient in the pond where temperature quickly changes from warm to cool. People often feel this when diving down into the water while swimming. When a sudden temperature change occurs following a storm or cold front, the epilimnion will cool down and begin to mix with the hypolimnion. This results in what is called a “turnover” as the surface water becomes cooler and denser and moves to the bottom of the pond while the colder water at the bottom is pushed upwards. This colder water has very little dissolved oxygen and can cause a fish kill, though this is not always the result. One preventative measure is installing a bottom diffuser to the pond. This device pumps air from the bottom of the pond to the top, which not only oxygenates the pond but also eliminates the stratification.
Total Dissolved Solids
Total dissolved solids, or TDS, is the sum of all the chemical ions dissolved in the water, often consisting of minerals and salts. TDS is strongly influenced by the surrounded geology. For example, granite is an inert substance that does not release ions and won’t dissolve easily. Therefore, TDS will be low in areas where the geology consists of granite. Land use activities and stormwater runoff can also influence TDS levels. As stormwater runs off the landscape, it can pick up material such as sediment, nutrients, and animal waste. TDS can be measured by a handheld meter and is measured in parts milligrams per liter (mg/L) or through a laboratory test. According to the Environmental Protection Agency, drinking water should have a maximum TDS of 500 mg/L. When TDS reaches a concentration greater than 500 mg/L but less than 3,000 mg/L it is considered freshwater. A reading of 3,000 – 10,000 mf/L is considered brackish, and anything above this threshold is saline.
As previously mentioned, the Clemson Agriculture Service Laboratory can provide values to numerous components of a pond. These include pH, salinity, phosphorus, potassium, calcium, magnesium, zinc, copper, manganese, iron, sodium, chloride, boron, sulfur, nitrate nitrogen, bicarbonate, carbonate, and the sodium adsorption ratio. For more information on how to submit a sample, see the Ag Service Lab Irrigation Water site.
Originally published 05/20




